Is It Safe to Use Flonase (or Other Nasal Steroids) During Pregnancy?
Rhinitis is characterized by congestion, which usually involves a stuffy, dripping nose and coughing. There are two types of rhinitis: allergic rhinitis and non-allergic rhinitis. (Learn More)
Allergies are common during pregnancy, and pregnant women may respond to allergens differently than they normally do. Some experience pregnancy rhinitis, or non-allergic rhinitis. (Learn More)
Flonase is used to treat rhinitis from seasonal and year-round allergies. (Learn More)
As with many drugs, there is limited research available about the safety of Flonase during pregnancy. However, a recent research analysis indicated that intranasal corticosteroid sprays like Flonase are likely to be considered safe if taken at proper doses as prescribed by a doctor after a full medical evaluation. (Learn More)
Discussing your allergy symptoms, along with any concerns about medications, with your doctor is the best option for finding an effective treatment during pregnancy. (Learn More)
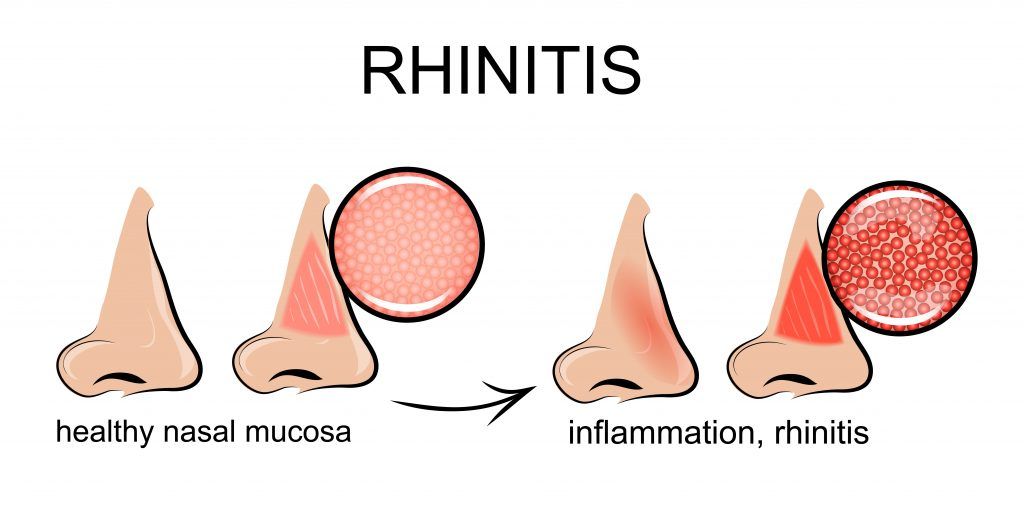
Understanding Rhinitis
Rhinitis is a condition that causes the lining of the nose to become inflamed or irritated. With allergic rhinitis, the nasal irritation can be linked to a certain allergy. Allergic rhinitis occurs when the body has an allergic reaction and in response, releases substances — histamine and leukotriene — that irritate the nasal area and lining. This results in swelling. With non-allergic rhinitis, the cause of the body’s reaction, which mimics an allergic reaction, is unknown.
Pregnancy and Rhinitis
Many women experience allergies during their pregnancy, including some who have never experienced seasonal allergies (sometimes called hay fever) or year-round allergies before. Those who have always experienced allergies may find their symptoms worsening with pregnancy, especially around the third trimester. Many pregnant women also experience pregnancy rhinitis. This may be labeled as rhinitis of pregnancy or simply non-allergic rhinitis. While the occurrence of non-allergic rhinitis in expecting women can’t be connected to a specific food or substance causing the nasal reaction, it is thought that the increase of hormones and body fluid may be related to increased sensitivity. Hormones associated with pregnancy have been shown to have both direct and indirect impact on nasal blood flow and nasal mucous glands. Rhinitis affects approximately 18 to 42 percent of pregnant women. The following are symptoms:
Sneezing
Nasal irritation
Running nose and congestion

What Is Flonase, and How Does It Work?
Flonase is a brand-name fluticasone nasal spray that treats symptoms of seasonal and year-round allergies. As a corticosteroid (also called a steroid or nasal steroid), Flonase reduces and prevents allergic rhinitis and different types of nasal inflammation. By lessening swelling and inflammation in the nasal area, Flonase alleviates some of the uncomfortable symptoms of allergies, including nasal congestion, sneezing, stuffiness, and a runny and/or itchy nose.
Is Flonase Safe During Pregnancy?
As with many drugs, there is limited research available about the safety of Flonase during pregnancy. Most women and drug companies are unwilling to risk the health of babies to test medicines. A 2018 research analysis indicated that intranasal corticosteroid sprays like Flonase were likely to be considered safe for pregnant women if taken at proper doses as prescribed by a doctor after a full medical evaluation. It also concluded that insufficient clinical research was available to fully prove or disprove these findings. According to the American College of Allergy, Asthma, and Immunology (ACAAI), intranasal corticosteroid sprays should be considered for pregnant women whose nasal allergy symptoms are considered more than mild and last more than a few days, as sprays may be a safer alternative to oral allergy medications. Along with this recommendation, ACAAI acknowledges that there is limited clinical research on the use of intranasal corticosteroid sprays during pregnancy. When the Food and Drug Administration (FDA) used a pregnancy grading system for labeling, Flonase was categorized as a Pregnancy Category C drug. Category C drugs are categorized as such if they have potential benefits for the expecting mother that warranted use during pregnancy despite potential risks. Generally, Category C drugs showed adverse effects on fetuses in animal studies but had not been studied adequately on humans. Ultimately, the FDA found the pregnancy letter category system to be overly simplistic as well as easily misinterpreted. They replaced it with the Pregnancy and Lactation Labeling Rule (PLLR).
Speak With Your Doctor About Flonase During Pregnancy
With most relatively safe medicines and drugs, use during pregnancy will likely come down to a consideration of risk and benefit. If a nasal flare-up is greatly affecting your well-being and causing you stress, your doctor may consider Flonase or another medication to be warranted, with the benefits of taking it outweighing the minor risks of use. When considering the continued use of an allergy medication during pregnancy or a medication to treat a new allergy problem that’s occurred during pregnancy, have an honest and thorough conversation with your doctor about your symptoms and concerns. This will be your best option to find relief and peace of mind.
References
Sinus Allergy Rhinitis Affects Millions. (January 24, 2013). Everyday Health.
What Is Flonase (Fluticasone)? (June 10, 2016). Everyday Health.
Allergies During Pregnancy. (April 29, 2019). What to Expect.
Congestion and Other Nasal Symptoms During Pregnancy. (March 31, 2019). Verywell Health.
Asthma and Allergic Diseases in Pregnancy: A Review. (March 2, 2009). World Allergy Organization Journal.
Natural Ways to Clear Up Rhinitis of Pregnancy. (April 7, 2016). Healthline.
Pregnant Women in Clinical Trials: FDA Questions How to Include Them. (April 9, 2018) MD Edge.
Safety of Intranasal Corticosteroid Sprays During Pregnancy: An Updated Review. (February 27, 2018). Official Journal of the European Federation of Oto-Rhino-Laryngological Studies (EUFOS).
Pregnancy and Allergies. (April 17, 2018). American College of Allergy, Asthma, and Immunology (ACAAI).
Flonase (fluticasone propionate). (April 29, 2003). U.S. Food and Drug Administration (FDA).
FDA Pregnancy Categories. (May 29, 2008). U.S. Department of Health and Human Services.
The Pregnancy and Lactation Labeling Rule (PLLR). September 14, 2016. U.S. Food and Drug Administration (FDA).

Related Articles

Allergies and Vertigo (Dizziness): Why It Happens and What to Do
If you have allergies, you may be dealing with several...
What’s the Most Effective Way to Treat Shingles? Can It Be Done Naturally?
If a rash has recently appeared on your body, you may...

Getting Over Performance Anxiety: What Works and What Doesn't?
Performance anxiety is a problem many people struggle...

Guide to Medication for Those Transitioning
People who are transgender have a different gender...
